|
9/11/2023 0 Comments Zn element ph Conversely, when the nutrient-type element concentrations are known, incorporation of Zn in foraminiferal shells can be used to reconstruct past sea water carbon speciation. The micro algal cell wall has indeed the capacity to bind with these ions via its negatively charged moieties 5. Our results suggest that foraminiferal Zn/Ca might be especially useful when combined with other proxies, enabling reconstruction of past seawater element concentrations. uptake ofof thethe element element from fromnutrient nutrient. mention that the pH and the cell’s wall play by themselves one of the most important functions in the adsorption of heavy metals by micro-algal cells. By modeling activities of different chemical species of Zn in seawater over a range of, we suggest that Zn2+, rather than other relatively abundant Zn-species (e.g. Here we show that zinc incorporation in cultured specimens of the benthic foraminifer Ammonia tepida is correlated to changes in carbonate ion concentration (). This work paves a way for designing high-performance organic electrode materials. Culturing benthic foraminifera under controlled carbonate chemistry conditions allows for disentanglement of impacts of different parameters of the carbon system on the elemental composition of foraminiferal calcite. 2 Ph electrode is demonstrated to accomplish dominated proton insertion from weakly acidic zinc electrolyte by comprehensive tests. high HCO3- concentration because of reducing conditions in. Still also nutrient-type elements will be most likely influenced by factors other than their relative concentrations. Zn is precipitated as sparingly soluble Zn(OH)2 when pH increases in acid soil following flooding. Zinc (Zn) is one of the most profusely abundant transition elements in the Earths crust and is also reported to be an essential trace element for all. Veroorzaakt ernstige brandwonden en oogletsel. wikipedia H-zinnen H 272-314 Kan brand bevorderen oxiderend. 
read more useful tools to reconstruct biogeochemical cycling and past ocean circulation. Formule: HNO 3 Molecuulmassa: 63,01 Plaats in de stock: 45. Whereas conservative elements incorporated in foraminiferal shell carbonate reflect factors such as temperature (Mg), carbonate chemistry (B) and salinity (Na), the nutrient type elements (Ba, Cd, and possibly Zn) are. This work expands the knowledge on zinc electrode kinetics over a wide range of pH and augments the existing literature on zinc corrosion.The trace elemental composition of foraminiferal shell calcite is known to reflect the environment in which the shell was precipitated. In alkaline conditions corrosion occurs chiefly by the formation of zinc hydroxide complexes or zinc oxides that could protect the surface depending on local pH and potential at the metal surface. 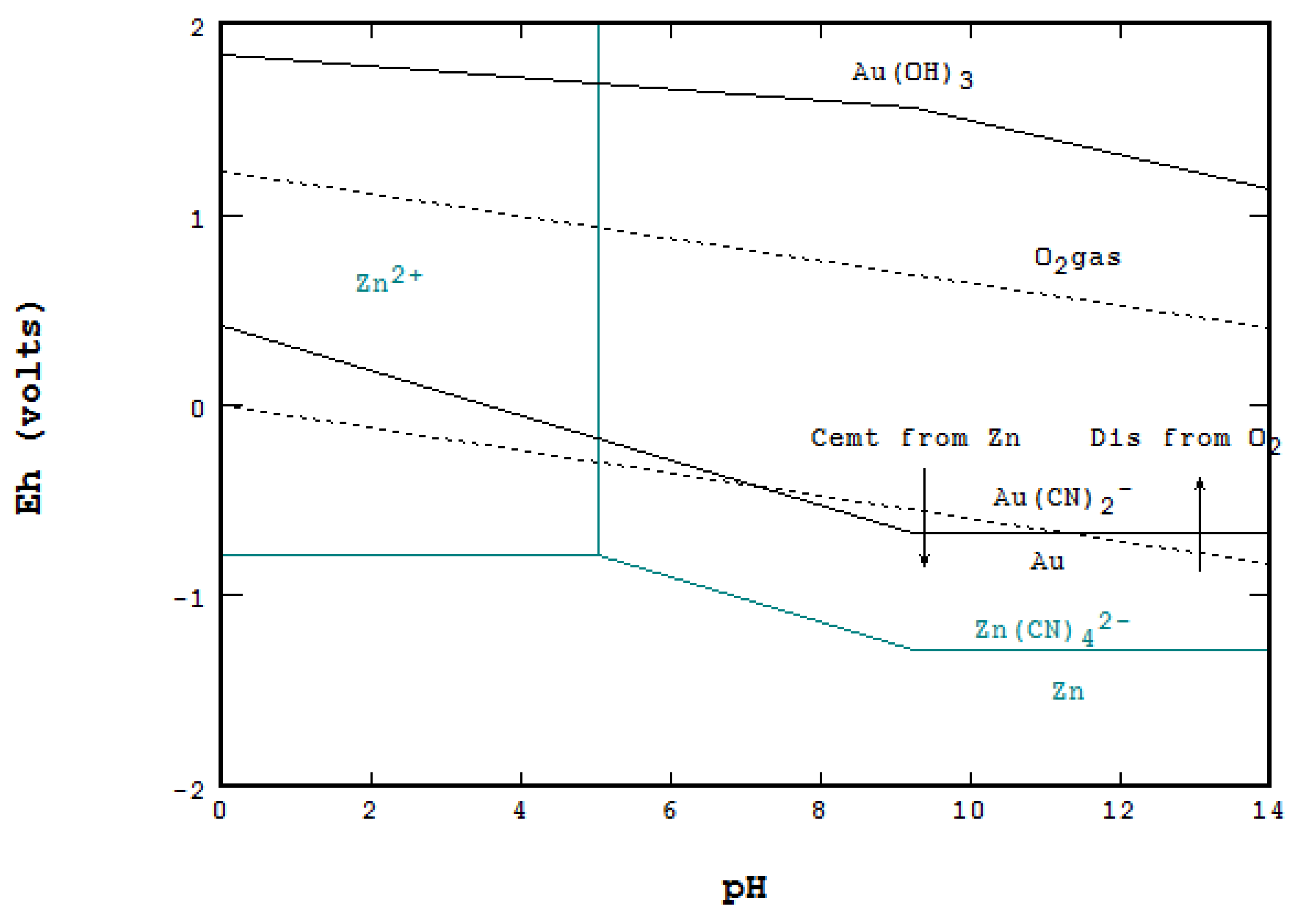
The surface oxides thermodynamically predicted to form in this pH (7 to 10) do not form an effective corrosion protection barrier. In the pH range (7 to 10), the lowered cathodic reaction rate reduces overall zinc corrosion rates. This range is illustrated by the degree of trace-metal pyritization studied by Huerta-Diaz and Morse (1992). These elements show varying tendency to be incorporated into pyrite. Local concentrations can be far above this range due to high Zn in the soil. These concentrations are higher than pristine concentrations due to historical enrichments (e.g. In acidic solutions, the active form of corrosion occurring is controlled by the kinetics of the cathodic reaction (predominantly hydrogen evolution). The authigenic precipitation is conditioned by alkalinity, pH, Eh and bacterial activity (Reimers et al., 1996. Zinc (Zn) is the 24th most abundant element in the earth, and is present in all soils in typical background concentrations 10100 mg Zn kg 1. The analysis shows that, in general, variations in corrosion of zinc with pH are associated with the cathodic currents. 
The electrochemical data was then contrast to thermodynamic chemical equilibrium diagrams. Soil pH has a dominant effect on solubility and therefore availability and potential phytotoxicity of ions (nutrients as well as toxic elements) (Clark and. Anodic and cathodic polarization of zinc was carried out in 0.1 M sodium chloride (NaCl) solution with pH ranging from 1 to 13. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
